Current Research
Sensemaking in Chemistry Titrations: Traditional vs. Vernier-Assisted Methods
In my AP Chemistry classroom, I am currently conducting research examining how laboratory tools influence students’ conceptual understanding of acid-base titration.
Titration is a foundational chemistry experience. Students collect quantitative data, calculate unknown concentrations and determine equivalence points and Kas. However, many students struggle to connect what they see in the lab (color change, a pH curve) with what is happening at the molecular level during neutralization reactions.
This study investigates a central question:
Does digital data collection technology enhance or hinder students’ scientific sensemaking?
Two intact AP Chemistry sections complete the same titration unit using different laboratory approaches:
Traditional method: Burette, phenolphthalein indicator, manual endpoint determination
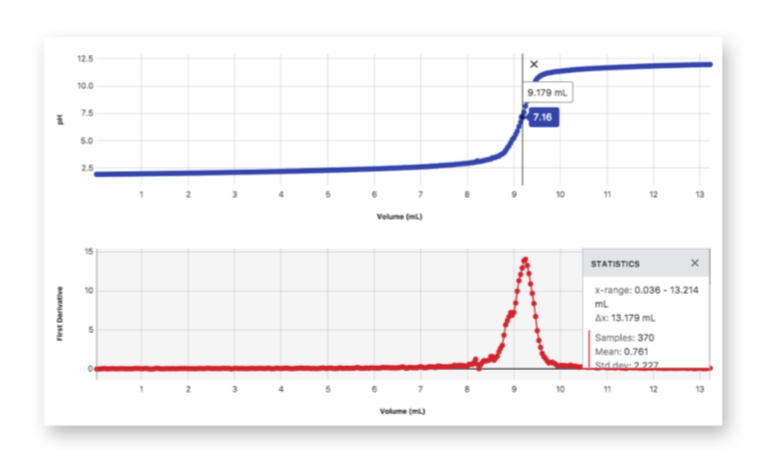
Vernier-assisted method: LabQuest 3, pH probe, real-time titration curve, automated equivalence analysis
Students complete pre- and post-assessments, lab reports, and reflective reasoning tasks. A subset of students participates in think-aloud sessions to capture real-time interpretation of titration data.
The study examines:
Conceptual gains in molecular-level reasoning
Accuracy and efficiency of titration results
Students’ ability to interpret graphical data
Differences in mechanistic explanations across conditions
Preliminary findings suggest that digital tools enhance data visualization and graph interpretation while traditional methods may strengthen procedural awareness and uncertainty reasoning. The goal is not to replace one method with another, but to better understand how instructional technology can be integrated intentionally in ways that deepen rather than dilute conceptual understanding.
This research contributes to broader conversations in chemistry education and instructional design about the role of technology in supporting authentic scientific reasoning.